|
3/31/2024 0 Comments Molar mass on period table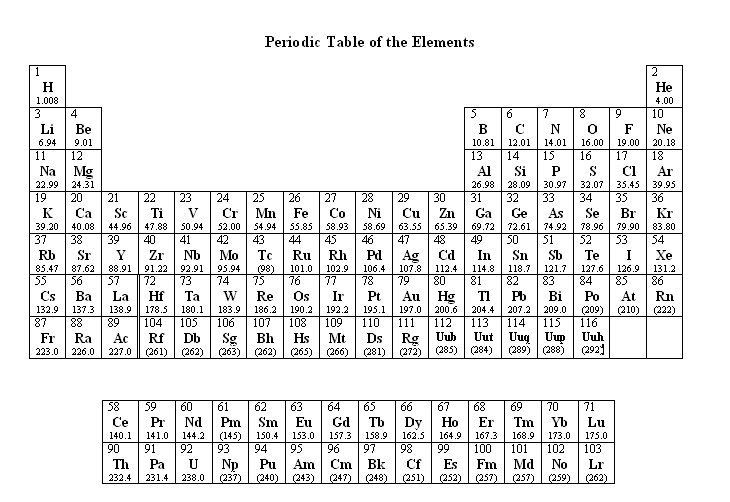 
The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. M = 58.7 + 2 x 35.5 + 6 x 18.0 = 237.7 g/molĬalculating the molar mass is essential as it lays the foundation for most of the calculations in stoichiometry such as the mass, moles, number of particles and etc.\). The periodic table lists the atomic mass of carbon as 12. M (NiCl 2♶H 2O) = M (Ni) + 2 M (Cl) + 6 M (H 2O) The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. He formula of the hydrate is NiCl 2♶H 2O, and therefore, the molar mass would be: To calculate the molar mass of a hydrate, determine the molar mass of the salt and add to the molar mass of the water multiplied by its subscript.įor example, calculate the molar mass of nickel (II) chloride hexahydrate. Then, lookup atomic weights for each element in periodic table: S: 32. M (NO 3 –) = M (N) + 3 x M (O) = 14.0 + 3 x 16.0 = 62.0 g/mol Computing molar mass step by step First, compute the number of each atom in S: S: 1. The formula of nitrate ion is NO 3 –, therefore, the molar mass would be: It is a result of either losing or gaining an electron(s) which has negligible mass compared to atoms and molecules.įor example, what is the molar mass of the nitrate ion? In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. Remember, the number outside of parenthesis applies to all the elements in it. Periodic Table of Elements - The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. Next, we write the ions next to each other and add the correct subscripts:Īnd now, we multiply the molar mass of each element by its subscript. Element 43 (technetium), element 61 (promethium), and most of the elements with atomic number 84 (polonium) and higher have their atomic mass given in. In studying the periodic table, you might have noticed something about the atomic masses of some of the elements. You need to know the common polyatomic ions from where we find that sulfate is the SO 4 2- ion. Answer: (a) noble gas, (b) chalcogen, (c) alkaline earth metal, (d) alkali metal. M (PCl 5) = 30.1 + 5 x 35.5 = 207.6 g/molĪnother example: what is the molar mass of aluminum sulfate?įirst, we know that the aluminum ion has a 3+ charge. 
To calculate the molar mass of PCl 5, we multiply the molar mass of each element by its subscript: Go over naming covalent compounds to refresh some of the concepts here. 
The formula of phosphorous pentachloride is PCl5. If the formula of the molecule is not given, you will need to first determine it.įor example, determine the molar mass of phosphorous pentachloride. To calculate the molar mass of a molecule, we add the molar mass of each constituent atom by the corresponding subscript.įor example, the molar mass of water would be: Usually, we round off the molar mass to 1 or 2 decimal places, so for P, we’d use 123.9 or 123.90 for its molar mass. For example, to find the molar of P 4, we multiply 30.974 g/mol by 4: If there is a subscript in the symbol, then you need to multiply the molar mass by that number. And we can do this for all the elements in the periodic table. For potassium then, it is 39.098 g/mol which means one mole of potassium atoms weighs 39.098 g. What’s important for today’s topic is that numerically, the molar mass is equal to the average atomic mass of the given atom, so we can look it up in the periodic table. Organic tin compounds may be highly toxic. It is a silvery-white, soft, malleable and ductile metal that ressist oxygen and water but dissolves in acids and bases. The origin of the name comes from the Latin word stannum meaning tin. The smaller one is the atomic number which shows the number of protons, and the larger one is the average atomic mass: Relative atomic mass: 118.710 (7) Tin has been known since ancient times. So, how do we find the molar mass in the periodic table?įor example, there are two numbers given for potassium in the periodic table: 19 and 39.098. To calculate the molar mass of an atom or a molecule, we are always going to need a periodic table. The mass of one mole of atoms/molecules/ions is called its molar mass ( M) expressed in g/mol.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
